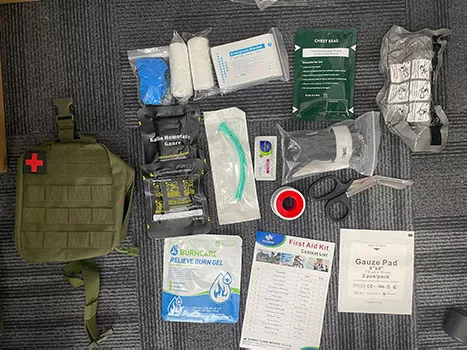
How We Ensure Quality in Bulk Tactical First Aid Kit Production
Introduction: Why Quality Control Matters More Than Price
For distributors, importers, and professional buyers, the biggest risk in sourcing a tactical first aid kit isn’t pricing—it’s inconsistent quality.
A single failed component—especially something critical like a tourniquet—can lead to:
- customer complaints
- product returns
- reputational damage
- regulatory issues
That’s why experienced buyers don’t just ask “what’s included in the kit?”
They ask:
“How is quality controlled across thousands of units?”
This is where most suppliers fall short.
Below is a transparent look at how quality is managed throughout bulk tactical first aid kit production—from component sourcing to final shipment.
Quality Starts Before Production: Component Selection
Quality issues rarely begin on the production line—they begin with poor component sourcing.
Not all components labeled as “medical” meet the same standards. For example:
- Tourniquets can vary significantly in material strength, windlass durability, and usability
- Gauze and bandages may differ in absorption, sterility, and packaging integrity
- Low-cost kits often substitute critical items with non-compliant or downgraded alternatives
To avoid this, a structured sourcing process is essential:
- Pre-qualified suppliers for each component
- Defined material specifications
- Consistency across batches
For bulk buyers, this ensures that the product you approve is the product you continue to receive.
Incoming Quality Control (IQC): Verifying Every Batch
Once materials arrive, they are not immediately used in production.
Each batch undergoes incoming quality control (IQC) to verify:
- material consistency
- physical integrity
- compliance with specifications
Depending on the component, this may include:
- visual inspection
- measurement checks
- random sampling
This step prevents defective or inconsistent materials from entering the assembly process—where issues become harder and more expensive to fix.
Standardized Assembly: Consistency at Scale
One of the most common concerns in bulk orders is consistency:
“Will every unit match the approved sample?”
This is addressed through standardized assembly procedures.
Key elements include:
- Clearly defined SOPs (Standard Operating Procedures)
- Structured workflow for each kit configuration
- Training for assembly staff on specific product requirements
Rather than relying on individual workers’ judgment, the process is system-driven.
This reduces variation and ensures that each tactical first aid kit is assembled exactly as specified.
Functional Testing: Ensuring Real-World Reliability
A kit may look complete—but that doesn’t mean it performs reliably.
For critical components, especially tourniquets, functional testing is essential.
This can include:
- usability checks (single-hand operation)
- tension and durability validation
- random sampling for performance testing
The purpose is simple:
To ensure that components don’t just meet visual standards—they function correctly in real-world scenarios.
For professional buyers, this is one of the most important indicators of product reliability.
Packaging Accuracy and SKU Control
In bulk supply, small errors can scale quickly.
Common issues in poorly managed production include:
- missing components
- incorrect kit configurations
- labeling mistakes
To prevent this, packaging control focuses on:
- SKU-specific assembly verification
- checklist-based packing processes
- label and barcode accuracy checks
This ensures that:
- each unit matches the intended configuration
- distributors receive exactly what they ordered
- downstream customers receive consistent products
Final Quality Inspection (FQC): Before Shipment
Before shipment, finished products go through a final quality inspection (FQC).
This step verifies:
- overall kit completeness
- packaging integrity
- consistency across the batch
Depending on the order size and requirements, inspections may follow defined sampling standards.
In addition, batch traceability is maintained, allowing any issue to be tracked back to its source.
For buyers, this adds a final layer of assurance before goods leave the facility.
Compliance and Documentation Control
For many markets, quality is not just about performance—it’s about compliance.
Different regions may require:
- CE-related documentation
- FDA-related considerations
- labeling and packaging standards
Managing this involves:
- maintaining proper documentation
- aligning product specifications with market requirements
- preparing export documentation for smooth customs clearance
This reduces the risk of delays, rejections, or regulatory issues after shipment.
Transparency During Production: Working with Buyers
One of the biggest concerns for first-time buyers is lack of visibility:
“What happens after I place the order?”
A structured process helps address this:
- Pre-production sample confirmation
- Clear specification alignment before manufacturing
- Optional inspection reports before shipment
This level of transparency ensures that buyers remain informed and in control throughout the process.
Continuous Improvement: Learning from Every Order
Quality control is not a one-time system—it’s an ongoing process.
Over time, improvements are driven by:
- customer feedback
- internal defect tracking
- refinement of production procedures
This allows the supplier to:
- reduce recurring issues
- improve consistency
- adapt to changing buyer requirements
For long-term partners, this means more reliable outcomes with each order.
Conclusion: Quality Is the Foundation of Reliable Supply
In bulk procurement, quality is not defined by a single inspection—it’s built into every stage of the process.
From component sourcing to final shipment, a structured quality system ensures:
- consistency across large orders
- reliable performance of critical components
- reduced risk for distributors and brands
For buyers, this translates into something more valuable than cost savings:
confidence in every unit delivered
If you are evaluating suppliers for tactical first aid kits:
- Request a sample to assess component quality firsthand
- Discuss your required specifications and market standards
- Review production and quality control processes before placing an order
❓ FAQs: Bulk Tactical First Aid Kit Quality Control
1. How can I verify the quality of a tactical first aid kit before placing a bulk order?
The most reliable way is to request a pre-production sample and evaluate:
- component quality (especially the tourniquet)
- packaging accuracy
- overall build consistency
You can also request documentation or inspection reports to understand the supplier’s quality control process.
2. What is the most important component to check in a tactical first aid kit?
The tourniquet is typically the most critical component.
Buyers should assess:
- durability
- ease of use (especially one-handed operation)
- material strength
Failure in this component can compromise the entire kit’s effectiveness.
3. What is IQC and why does it matter in first aid kit production?
IQC (Incoming Quality Control) refers to inspecting materials before production begins.
It matters because:
- defects are caught early
- only compliant materials enter assembly
- overall product consistency improves
4. How do suppliers ensure consistency across large orders?
Consistency is achieved through:
- standardized assembly procedures (SOPs)
- trained staff
- controlled workflows
This ensures every unit matches the approved sample.
5. What is included in final quality inspection before shipment?
Final inspection typically checks:
- completeness of the kit
- correct components
- packaging integrity
- batch consistency
This is the last step before goods are shipped.
6. Can I request third-party inspection for my order?
Yes, many buyers choose to use third-party inspection services for additional assurance.
This can be arranged before shipment to verify product quality independently.
7. How do I ensure the product meets my country’s regulations?
You should:
- confirm required certifications (e.g., CE, FDA-related compliance)
- verify labeling and documentation requirements
- communicate your market standards clearly to the supplier
8. What are the most common quality issues in bulk first aid kits?
Common issues include:
- inconsistent components
- low-quality substitutes
- missing items in kits
- incorrect labeling
These are typically the result of weak quality control systems.
